 
The emission of another particle was a probable explanation of this behavior, but searches found no evidence of either mass or charge. Another anomaly was the fact that the nuclear recoil was not in the the direction opposite the momentum of the electron. The electron antineutrino carries most of the remaining energy, the recoil energy of the nucleus being very small.Įarly studies of beta decay revealed a continuous energy spectrum up to a maximum, unlike the predictable energy of alpha particles. =(209.984095 u -209.982848 u)(931.502 MeV/u)įor beta decay, involving three particles, the beta electron energy is distributed from a low value up to the energy yield of the decay. The electron from the beta decay of 32P can approach the full decay energy of 1.71 MeV, but it has an average value of about 0.7 MeV.įor another numerical example, consider the example of the beta decay of bismuth 210Bi cited by Krane. It can be calculated from the difference in neutral atomic masses for 32P and 32S. The total measured energy release Q is consistent with the identified nuclear processes. The decay of 32P can be considered to be a collective beta decay process, examined in more detail above. The process of beta decay must be examined from the point of view of the binding energy difference between the parent and daughter nucleus, and the energy yield is then distributed to the products of the decay. Using the radius and charge, the electric potential energy of the electron would be KQe/r = -9.09 x 10 -15 J = -5.68 MeV! It is hard to conceive of an electron escaping that!Ĭonsidering that placing an electron at the radius is not plausible, look at the entire beta decay process from the point of view of the nuclear binding energies involved. Then the electric potential energy could be calculated.įor a mass number A = 32 for 32P, the nuclear size model yields r = 3.8 x 10 -15 m.įor 32P with 15 protons, the nuclear charge Q = 2.40 x 10 -18 coulombs. An estimate of the electrical binding energy if it could be placed at the surface of the nucleus may be made by treating the 32P nucleus as a sphere of charge and using the nuclear size model to calculate its radius. How Does the Beta Electron Escape All Those Protons?Įven though this is not plausible physically (e.g., the electron is not a point particle, it cannot be localized at such a point, etc.) it is an interesting conceptual question. 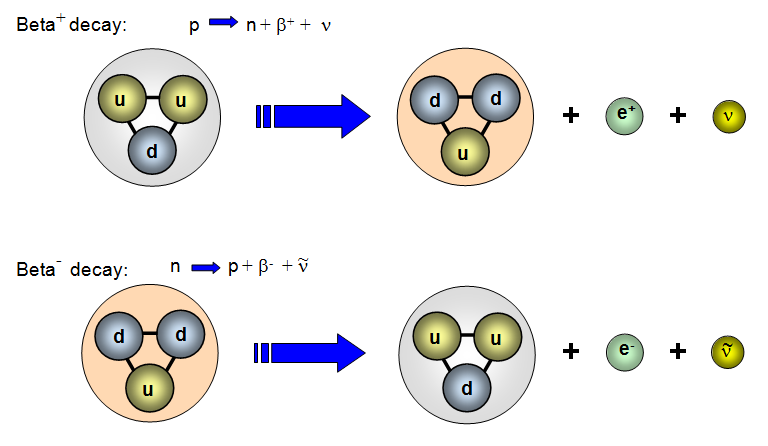
The use of a weak interaction Feynman diagram can clarify the process. Beta decay can be seen as the decay of one of the neutrons to a proton via the weak interaction. The emission of the electron's antiparticle, the positron, is also called beta decay. The radiation hazard from betas is greatest if they are ingested.īeta emission is accompanied by the emission of an electron antineutrino which shares the momentum and energy of the decay. The high energy electrons have greater range of penetration than alpha particles, but still much less than gamma rays. Beta particles are just electrons from the nucleus, the term "beta particle" being an historical term used in the early description of radioactivity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
